Warming Thoughts © #4-- A Deep Dive into DAC and the Beginning of a DAC Spider Chart
By: James F. Lavin, CEO Electron Storage, Inc.
Warming Thoughts © #4-- A Deep Dive into DAC and the Beginning of a DAC Spider Chart
By: James F. Lavin, CEO
Electron Storage, Inc.
Direct Air Capture (DAC) is an important and heavily promoted attempt to rectify global warming, with new startups being announced and established regularly around the world. DAC is part of a suite of approaches under the title Negative Carbon Emissions that span from growing trees, to growing seaweed to sink to the ocean depths, to accelerated mineralization by grinding ultramafic rocks, to ocean alkalization, to trapping CO2 in concretes. Due to limitations in scaling biological and other solutions, and because we need to remove more than 1,000 gigatons from the atmosphere, DAC remains a critical technology. There is no practical solution to alleviating global warming without DAC. Scaling DAC will take a miracle (and a lot of hard work and resources), so let’s get to work creating that miracle! This has been my attitude since I met Klaus Lackner in 2013
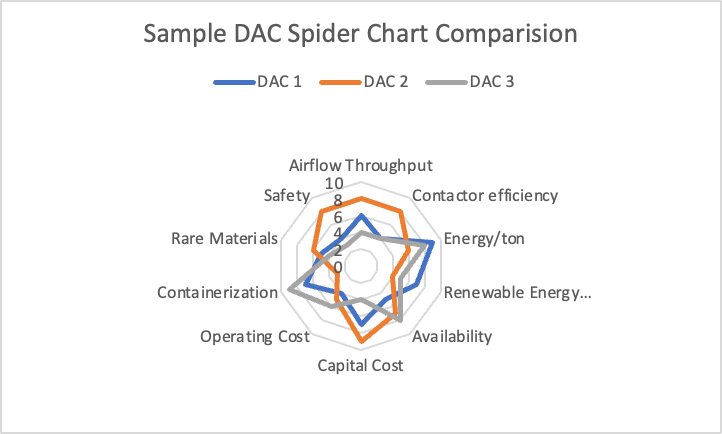
This Warming Thoughts will take a deep dive into various technological approaches to Direct Air Capture, capturing CO2 from out of the atmosphere around us. We will start to lay the basis for a spider chart, commonly used in battery technology evaluations, to quantify the strengths and weaknesses of various technologies along multiple different dimensions. Like batteries, with DAC there is no one best solution and many tradeoffs.
.
Ten years ago, DAC was still a fringe idea, primarily promoted by Professor Lackner, who is now head of ASU’s Center for Negative Carbon Emissions. Klaus, like many of the deepest thinkers on global warming, is a physicist. Nearly all physicists deeply understand thermodynamics. And thermodynamic limits create the fundamental problem with DAC – it requires lots and lots of energy. These physicists also think in gigajoules, which is a unit of energy 3.6x as large as megawatt hours, so 3.6 GJ equals 1 MWH. We will use both measures.
DAC Issues/Dimensions:
The first issue when considering DAC is airflow throughput. Fans, according to discussions I have had with Global Thermostat and Climeworks and papers from David Keith, peg the real-world energy for moving air at roughly 10% of the total energy requirement. This is not insignificant, particularly when you realize the energy needed by Climeworks’ capture plants is as high as 10GJ/ton, which means around 1 GJ for fans alone. The problem is that you need 1.3 million cubic meters of air, around the volume of a large enclosed stadium for a single ton. Since DAC is generally striving for economic efficiencies of ½ - ¾ of the CO2 being removed, that means moving more than 2 million cubic meters of air, or two stadiums, into contact with a capture medium, per ton. This problem is compounded when trying to move air through tightly packed sheets, a 3d capture medium, or a counterflow (liquid down/air up) column for liquid absorbants. These all require considerable air pressure to overcome air resistance.
Another factor tied to airflow is the aggressiveness of the capture medium. Simply put, the stronger and faster the reaction between CO2 in the air and the capture medium, the faster one can move air through it and the less total surface area of contactor required. Several capture companies use batch processes in which air is pushed into a chamber, captured over a period of minutes to hours, then the CO2 is exhausted from the chamber through some combination of heat and moisture (often steam for both), and vacuum. A batch process is going to have less throughput than bubbling air through a caustic soda liquid medium or across an aggressive electrochemical membrane. Throughput of air is therefore one of the key dimensions in evaluating DAC methodologies.
The second issue is the area of the contact medium. At one extreme is a company that plans to use finely crushed ultramafic rocks to create a vast surface area and then lets natural winds move over them without fans. They then heat the rocks to expel the CO2 and put them back out again. At the other extreme is the development of electrochemical charged membranes that attract CO2, or CO2 with some moisture, and then release the CO2 once a chamber is evacuated from air, or alternatively selectively pass the CO2 when the proper electricity is applied. In either case, the you need to create vast areas with specialized membranes inside chambers and support structures, and get them to react fast enough with air molecules moving at a significant speed.
The essential problem is that air isn’t very dense, and the membrane is essentially 2d, even if honeycombed and it has a limited surface area, and/or difficulty with getting effective low energy air penetration into the 3d structure. This is fundamentally true whether there are multiple thin layers, or honeycomb structures; you still have a presumably expensive solid structure trying to catch air molecules. Klaus Lackner has attempted to create mechanical trees by creating stacks of disks with a shag carpet-like material that attracts CO2 molecules when dry and rely on passing winds.
Liquid solutions of capture chemicals provide an alternative approach. Liquids can easily create fine droplets with tremendous surface area, or thin sheets with turbulent flow, providing a great deal of contact surface at a low cost using adapted cooling towers. Carbon Engineering uses this approach with an aqueous KOH solution and crossflow air direction. In addition to surface area, the great advantage of liquid droplets is that the process can be performed continuously. Most post-combustion (flue gas) carbon capture uses counterflow columns with liquid amine solutions such as MEA that are effective at capturing high concentration CO2 and good at releasing it at relatively low energies.
While all this sounds a little vague, the 1 Mt/year plant under development by Carbon Engineering will require a 20-meter-high wall that is 7 meters deep, primarily for the mixing of air and KOH solution, and will require around 40,000 square meters of total inlet area. If the wall is 75% inlet area, that means the wall will be 1.5 km long. To capture a gigaton/year, the wall would need to be 1,500 Km long, and still be 7 meters deep. And this is for one of today’s most efficient capture schemes!
The third issue is energy: The first phase of the DAC process is to grab CO2 from the atmosphere, which only contains 418 parts per million or around 4 black balls per 10,000 balls. One advantage of DAC vs. capturing gas from a power plant stack where it might be, say, 1,000 balls per 10,000 and therefore easier, is that we want to capture almost all the CO2 from the fossil fuel power plant. But from the air, it can be any economically ideal number of balls. Thermodynamically, there is a minimum energy of around .5 GJ/Ton for DAC. Less energy is needed in a concentrated stream from flue gas, but the need to capture almost all of the CO2 reduces this energy advantage.
Unfortunately, the theoretical minimum is just that, a theoretical minimum for a once through process. Creating a capture medium, whether it is mined and crushed rock, a sophisticated membrane, or a high PH solution, takes energy and raw materials. You also still have to devote energy and space to disposing it and making sure the capture is permanent. Except for carbon capture in mine tailings, perhaps enhanced with extra pipes to inject air under the surface, you need to regenerate your capture medium. It takes energy to induce the CO2 to release. (In most cases the fundamental carbonization reaction is exothermic, i.e., it releases energy, and the combined product is in a lower, more stable energy state. This means it takes energy to split the molecule and release the CO2.) Electrochemical membranes may operate in the opposite manner, using energy to bind the CO2. The release is free with this approach, but the need for energy to complete the cycle remains.
Energy for the three established DAC companies, as well as Heirloom who uses crushed rock, comes from heat, usually supplemented by vacuum or moisture/steam. Global Thermostat and Climeworks both use relatively low temperatures, near 100C, and therefore can claim low energy usage by using “waste heat.” This is especially the case for Climeworks’ initial plant located on top of a sewage treatment plan. Carbon Engineering, the only company seriously working on 1 Mt/year scale plants (still 1/1000th of a gigaton!), opted for high temperatures in their facility, around 900C, produced by natural gas with the CO2 emissions capture as part of the process. Heirloom also uses 900C heat.
It is important to note that total energy, not the temperature required, is what matters. Carbon Engineering’s process is reportedly significantly more energy efficient overall than the other DAC companies. The fundamental problem is that in chemical capture processes, you need to apply the energy to the entire mass of the capture medium, whether it’s membranes, the support structure, the moisture, a pellet, rock (carbonated or not), and the entire aqueous solution for liquid capture mediums. You also must pump or otherwise transport significant masses of your capture material, which of course requires energy as well.
Electrochemical membrane approaches are attractive due to the application of energy just to the membrane and the CO2 or CO2 containing molecules, without needing to heat extra masses. An alternative approach without heat or electricity comes from acid base chemistry. If CO2 is captured in a caustic soda such as KOH and mixed with acid, the CO2 will release. However, you still need energy to create the acid. There really is no free lunch.
The generally agreed upon theoretical minimum energy required for DAC is 1.1-1.2 GJ/Ton. There are no processes I am aware of that claim to be lower than 2 GJ/ton even under ideal conditions. In the real world with pumping losses, heat losses, and friction, anything less than 3GJ/ton would be great, and this is still less than 1 MWH/ton. By comparison, Carbon Engineering is projecting to be in the 6 GJ/ton when it builds its plant. Global Thermostat and Climeworks were reported to be in the 10-12 GJ/ton range, although there are claims this is now closer to 8 GJ/Ton. Energy Matters, particularly as this energy needs to be clean energy.
An additional, and non-trivial, energy requirement that applies to everyone is the compression of CO2 into a liquid state so it can be effectively transported or put into pipelines for underground disposal. This energy is approximately 120 KWH/ton. Depending on the process, another energy requirement may appear: the purity of CO2. Ideally, CO2 should be quite dry (to avoid carbonic acid formation) and at least 97% pure so nitrogen doesn’t interfere with underground mineral carbonatization. Further purification upon release from the capture medium may take considerable energy.
Before leaving the scene, an honorable mention must be given to Lackner’s mechanical trees. His amine-based capture coating develops an electrical charge when water evaporates from it’s surface. This charge enables it to capture CO2 at a modest rate, and when collapsed down into a tub of water, the CO2 discharges into the water, thus creating a solution of up to 10% CO2. The CO2 can be induced to bubble out, or just pumped into the ground along with the water. The wet membranes are then hoisted into the air again. This approach is brilliant from an external energy consumption basis (you still can’t beat thermodynamics, the sun’s heat and wind supply the needed energy). However, it too has limits. For instance, you need plenty of water in a dry climate, the CO2 solution is acidic, you still must concentrate the CO2 into a dry gas for most purposes, and you have thin membranes hanging in the wind and subject to dust and debris.
Compatibility with renewable energy sources is our fourth issue/dimension on the spider chart. How interruptible is the process? How well can power requirements be ramped down or quickly increased to stabilize the grid? As we discussed, capturing large amounts of CO2 with DAC requires massive amounts of energy. Using existing processes, capturing 10 gigatons a year will require 9-16% of the world’s energy. The true cost of renewable energy escalates dramatically if we assume it must meet grid requirements for voltage stability and constant power. Can the capture process provide grid services like rapid load ramp up to stabilize voltage, or can a process be reversed to provide power to the grid? How much energy is lost when power is cut off? If it’s a high heat process in addition to lost energy, does something cake or cause corrosion?
Electrochemical membrane processes seem to hold more promise of compatibility with a renewable grid, but leaving acidic vapor around (CO2+H2O means carbonic acid is formed) in a chamber may harm delicate membranes as do bubbles formed from high energy densities. In general, I think the evaluation of DAC, and carbon negative technologies in general, is too accepting of the availability of reliable grid electric power without considerating that the cost of such reliable power in a renewable world will be higher than our current fossil fuel-based power and may not be available at many locations.
The fifth dimension is availability: How long can a process operate before a shutdown for maintenance is required, and how long will such maintenance take? How continuous is the process? If using an electrochemical processes, how often do the membranes or electrodes need to be changed? How often do you need to replace pipes due to corrosion? If it’s a batch process, how long does it take for air to fill the chamber, or CO2 to be exhausted? Can it operate in winter storms with ice, or in the rain with high moisture levels, or in the heat of summer? What about dust storms that can introduce organics and silica into the process? How do you purge impurities from your recycled capture medium? How sensitive is a membrane to dust contamination? An increase in availability usually flows from operating experience, which is very limited with CO2 capture as it is an emerging industry, but inherent to many DAC processes are limitations on availability/duty cycle.
The sixth dimension is capital cost, or cost per ton of CO2 captured to build the plant. An exotic membrane may be very energy efficient and meet the criteria for interruptible power, and even be durable enough to meet availability requirements, but if it costs dramatically more than other solutions, it will be unaffordable. If the capital cost is high, the cost of interruptions rises, so it must have very high availability and low operating costs to be competitive. If capital costs are low, the project can sustain interruptions from renewable power and more maintenance downtime. Above all, at gigaton scales any solution will require 10’s of trillions of dollars of capital investment, so a 2x difference could impact world GDP. Capital costs matter.
The seventh dimension is operating costs: While energy requirements form a substantial part of operating costs, maintenance costs, part replacements, working material replacements, and number of people required to operate the plant all add to operating costs. LCOA, levelized cost of ownership, attempts to merge capital, longevity and operating costs into a single number. But at trillion-dollar levels, the sensitivity to discount rate, energy costs, and lifespan assumptions is too great to allow the assumptions to be concealed. Operating costs matter.
The eighth dimension is plant size/containerization: The more containerized the process, the greater the potential for a) cost reductions due to mass production, b) locational flexibility to minimize the cost of energy inputs, and c) the maximum potential to locate near geology that allows permanent CO2 disposal or, conversely, CO2 utilization in renewable fuels or other products. How much of the guts of the plant can be fit into containers? Due to airflow considerations, the contactor area is going to be the most difficult to containerize, but here too this dimension illuminates important differences. The more reactive a capture agent, the faster the airflow over it can be and the smaller the size of the contactor. Size matters.
The ninth dimension is the use of rare materials. Ruthenium is a wonderful catalytic agent to enhance various electrochemical reactions. It also costs $20,000 a kilogram, and the total world output is only around 35,000 kilograms and largely comes from Russia. Producing hundreds of square kilometers of a membrane coated with Ruthenium is going to be extremely difficult. Every DAC process needs be evaluated against critical inputs of rare materials if the process is going to be capable of achieving serious scale.
The tenth dimension is safety. Some processes are going to be inherently safer than others. Crushed rock itself may be benign but moving it may not. Natural gas and high heat or steam create dangers. MEA is a nasty substance. Ammonia is hazardous. Hydrogen is going to increase safety risks, as are strong acids or bases. When contemplating gigatons, safety matters.
In creating a spider chart, some of these factors may be qualitative, but many such as capital costs/ton, operating costs/ton, and energy usage/ton are not and can be scaled. We believe a spider chart analysis of all DAC methodologies will be helpful.